We Don't Do Pest Control. We Engineer Compliance Programs.
SafeCore was built on a simple premise: regulated facilities in New Jersey deserve pest management programs designed by people who have actually walked audit floors, not generic pest control companies learning on the job. Our methodology draws from over 20 years of designing and implementing IPM programs in food processing, pharmaceutical, and personal care manufacturing environments — across multiple countries and regulatory frameworks. That experience is what separates a compliance-grade program from a spray-and-pray service.
SafeCore exists specifically for these environments. Our programs are designed by professionals with ACE certifications, PCQI training, and deep knowledge of HACCP, GMP, and third-party audit standards — people who understand what "audit-ready" actually means in practice.
Every program starts with your regulatory reality — the standards you're held to, the audits you face, and the documentation your quality team needs.
Compliance-First Thinking
Every recommendation evaluated through a compliance lens before it reaches your facility.
Transparency & Accountability
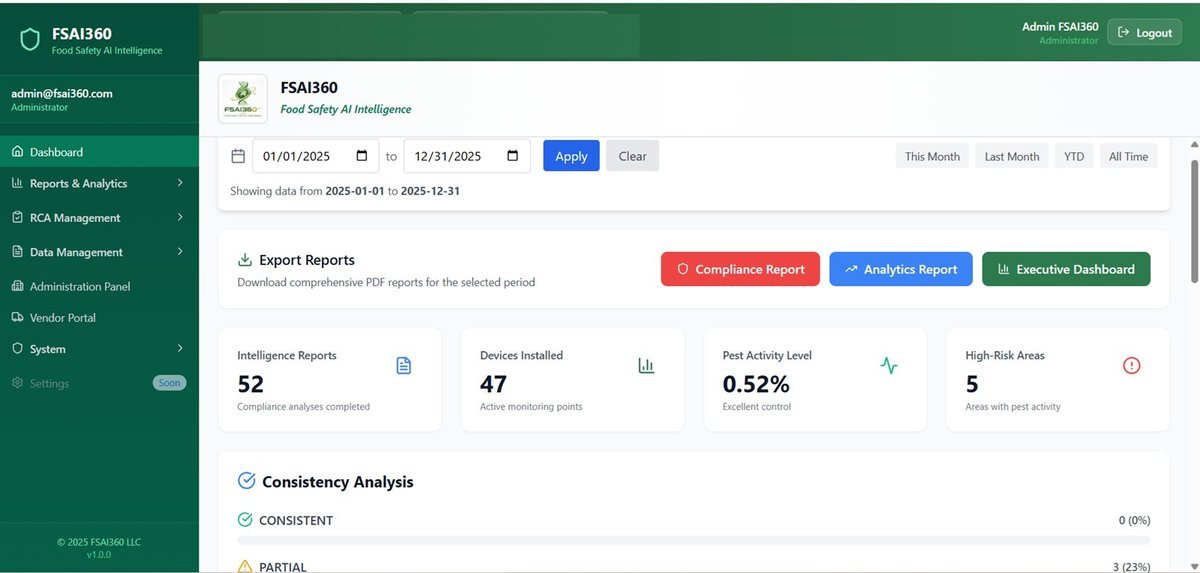
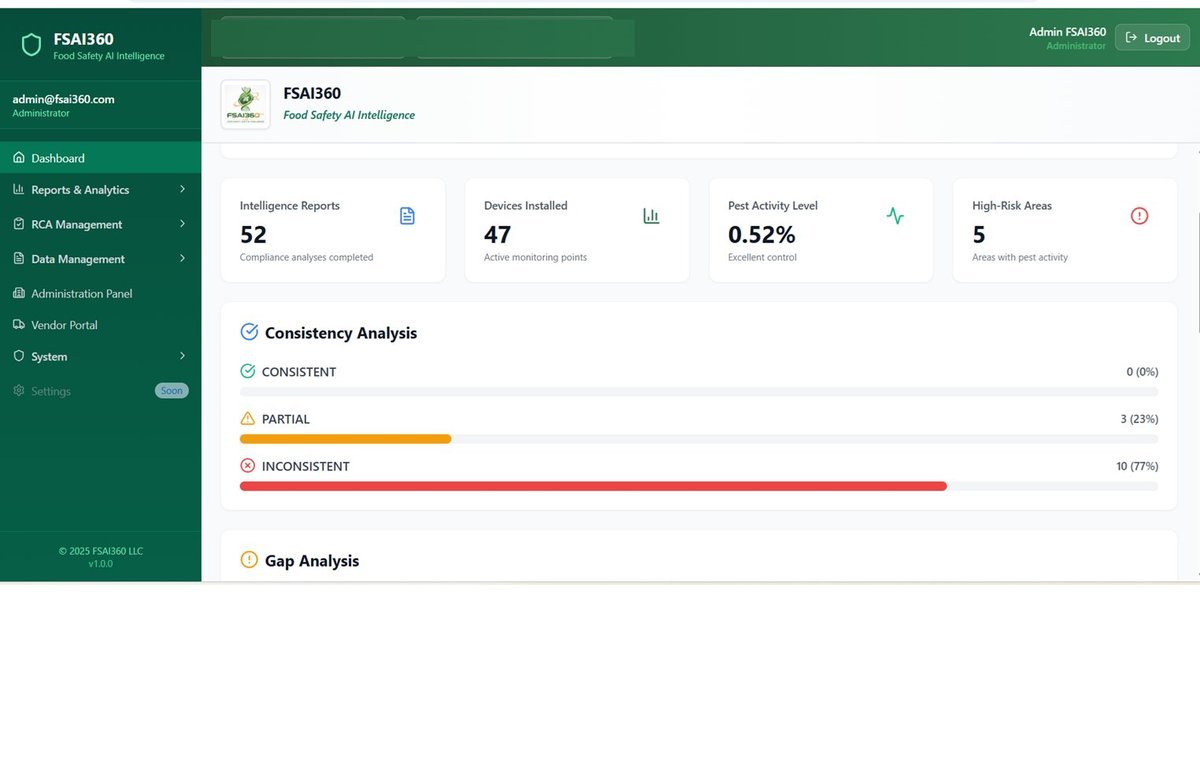
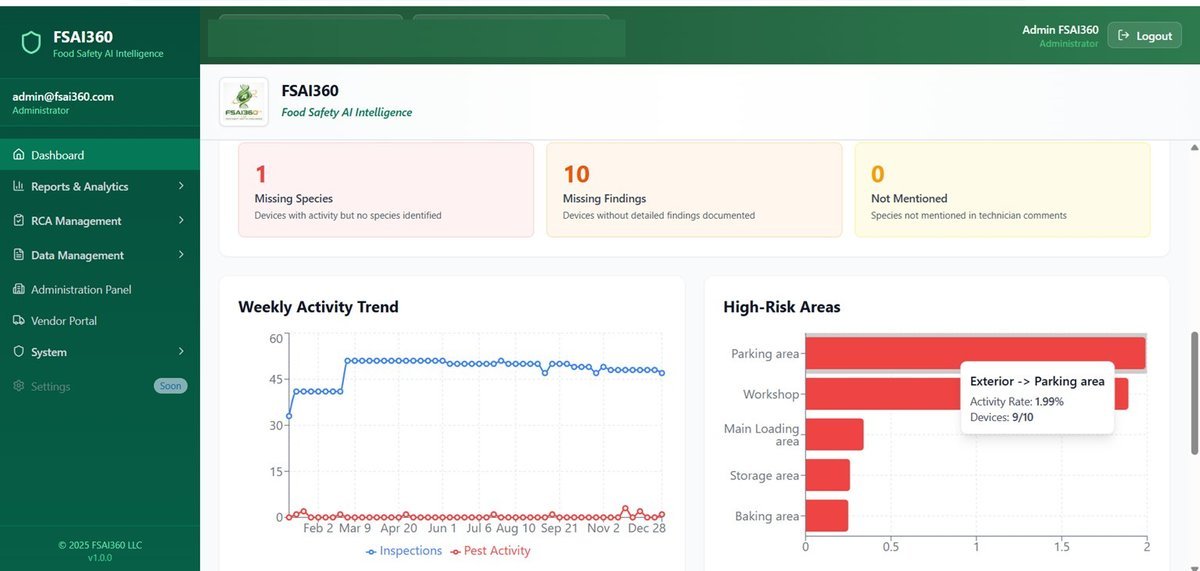
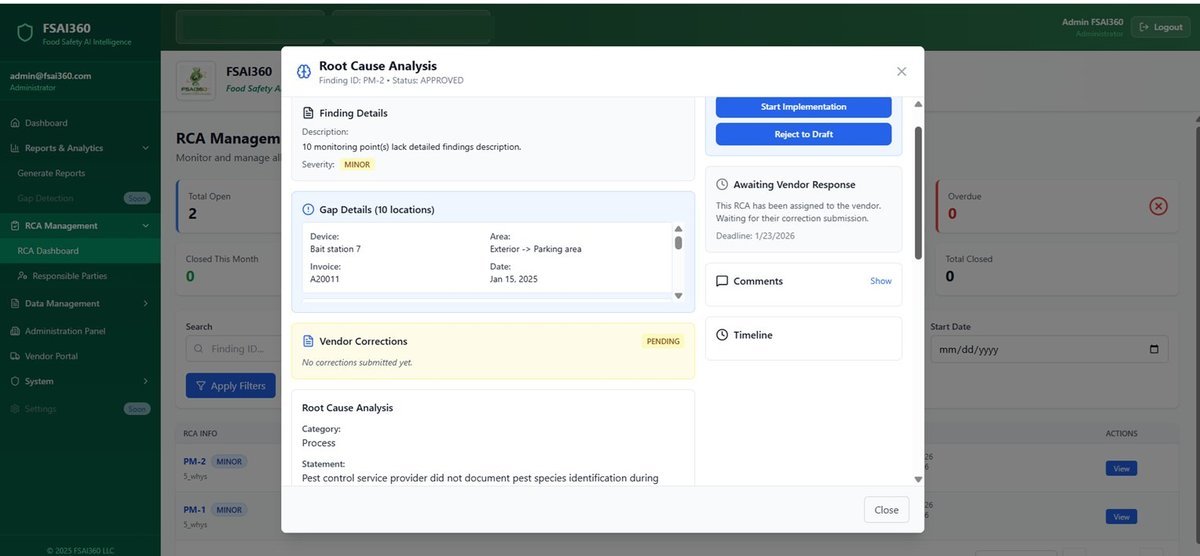
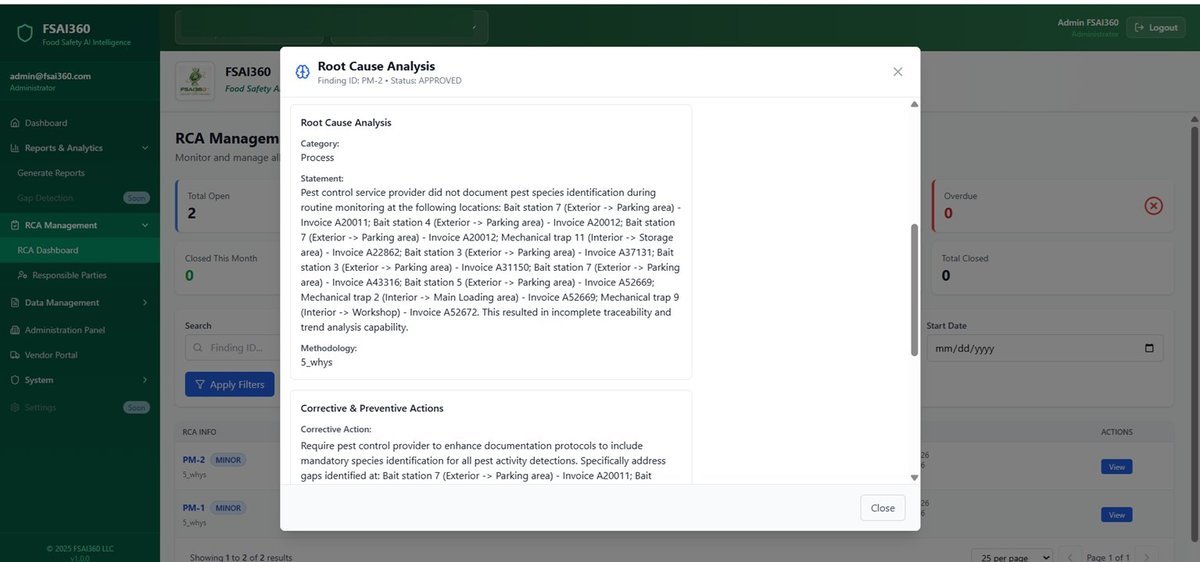
No hidden findings, no vague reports. Full real-time visibility through FSAI360.
Continuous Improvement
Built-in review cycles and data-driven adjustments keep your program evolving.